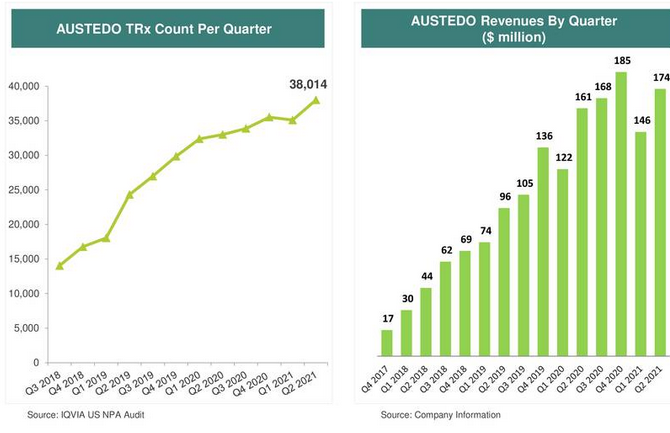
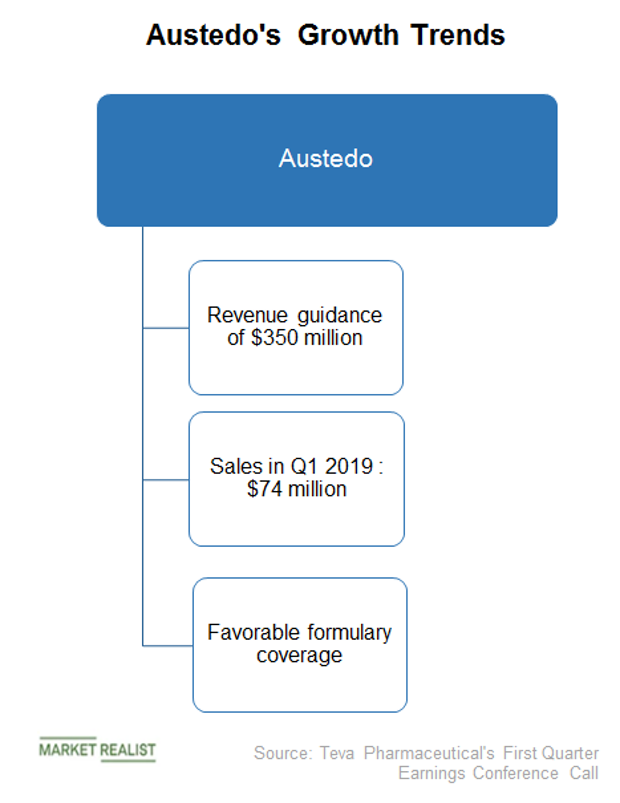
Teva Announces FDA Approval of AUSTEDO™ (deutetrabenazine) Tablets for the Treatment of Chorea Associated with Huntington's Disease

Teva's Austedo, struggling from a pandemic slowdown, needs huge gains to meet 2021 projections | Fierce Pharma

Teva announces FDA approval of Austedo tablets for chorea associated with huntington's disease - Pharma Advancement

Yo-Bio on Twitter: "$TEVA #Austedo have no black box warning for tardive dyskinesia. Only for HD (Read about HD and you will understand way). $NBIX #INGREZZA https://t.co/lXnoHCEUAA" / Twitter

Teva launches first DTC for Austedo into competitive tardive dyskinesia 2-drug market | Fierce Pharma